WHAT IS ISO 13485 ISO
The more specific requirements for validation of systems such as Enterprise Resource Planning (ERP), QMS and Laboratory Information Management Systems (LIMS), as well as any other applications used in the development or maintenance of medical devices are in ISO 13485.

One of the best ways to do so is to have Key performance indicators for the processes within the QMS.Ĭompanies can demonstrate that they are serious about protecting safety and quality by validating their computer systems. During the continuous monitoring of the QMS for its effectiveness, adjustments should be made as necessary and documented. There should be defined roles and responsibilities in documented quality agreements with any resources that are outsourced. Monitoring and controlling processes that impact the requirements of the risk-based approach: Whether you run the processes in-house or outsource some of them, processes that impact requirements of the risk-based approach ISO 13485:2016 must be monitored and controlled. While addressing the specific regulatory requirement of the market where the medical device is to be manufactured and marketed, the QMS content should also address the requirement of the ISO 13485:2016. The regulatory requirements vary across markets. Quality record documentation is performed to show that the QMS is executed and followed. It also provides the sequences, interactions and resources required to do business with a medical device company. What QMS consists of: The QMS consists of the fundamental set of policies, procedures, forms, and work instructions. The following section will take you through the major sections of the ISO 13485:2016 and provide action items and best practices to help you apply them in your medical device company.ĭefinition: "A formal system that documents the structure, processes, roles, responsibilities and procedures required to achieve effective quality management." (ASQ, a Quality Management System). There is a greater emphasis on risk-based approaches to quality and safety as compared to the ISO 13485:2003.Ĭompliance with ISO 9001:2016 requires companies to demonstrate that risk is taken into account for the entire organization's Quality Management System (QMS) processes that include aspects shown in the following figure.Ĭompliance with ISO 9001:2016 requires companies to demonstrate that risk is taken into account for the entire organization's Quality Management System (QMS) processes However, it requires risk-based approaches for quality and safety protection just like the requirement that is mentioned over a dozen times in the ISO 9001:2016 standards. The ISO 13485 standard does not have a high-level Annex SL structure that's found in the other ISO 9001:2015 standards.

WHAT IS ISO 13485 UPDATE
The update to ISO 13485:2003 helps in aligning with the regulatory requirements with respect to Documentation, Complaint management, and reporting issues to regulators. Many Global Regulatory bodies are including ISO 13485 into their requirements making the product suitable for companies to market internationally. Why ISO 13485 is revised and approved in 2016?.
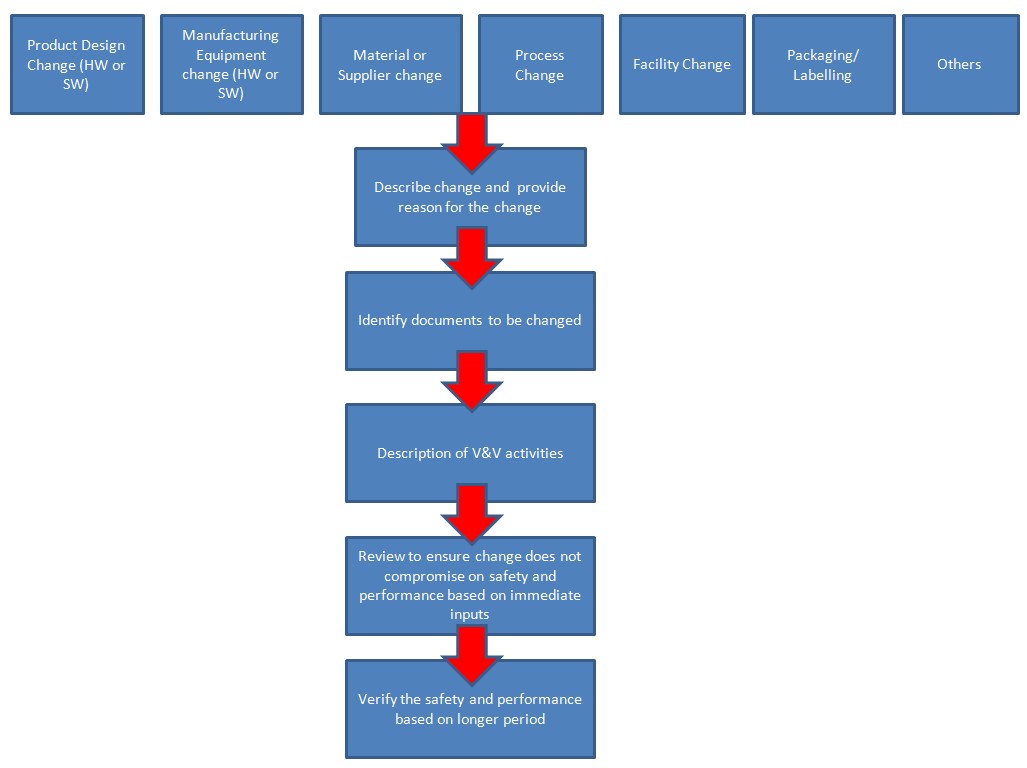
This article provides an overview of the major sections of ISO 13485:2016.
